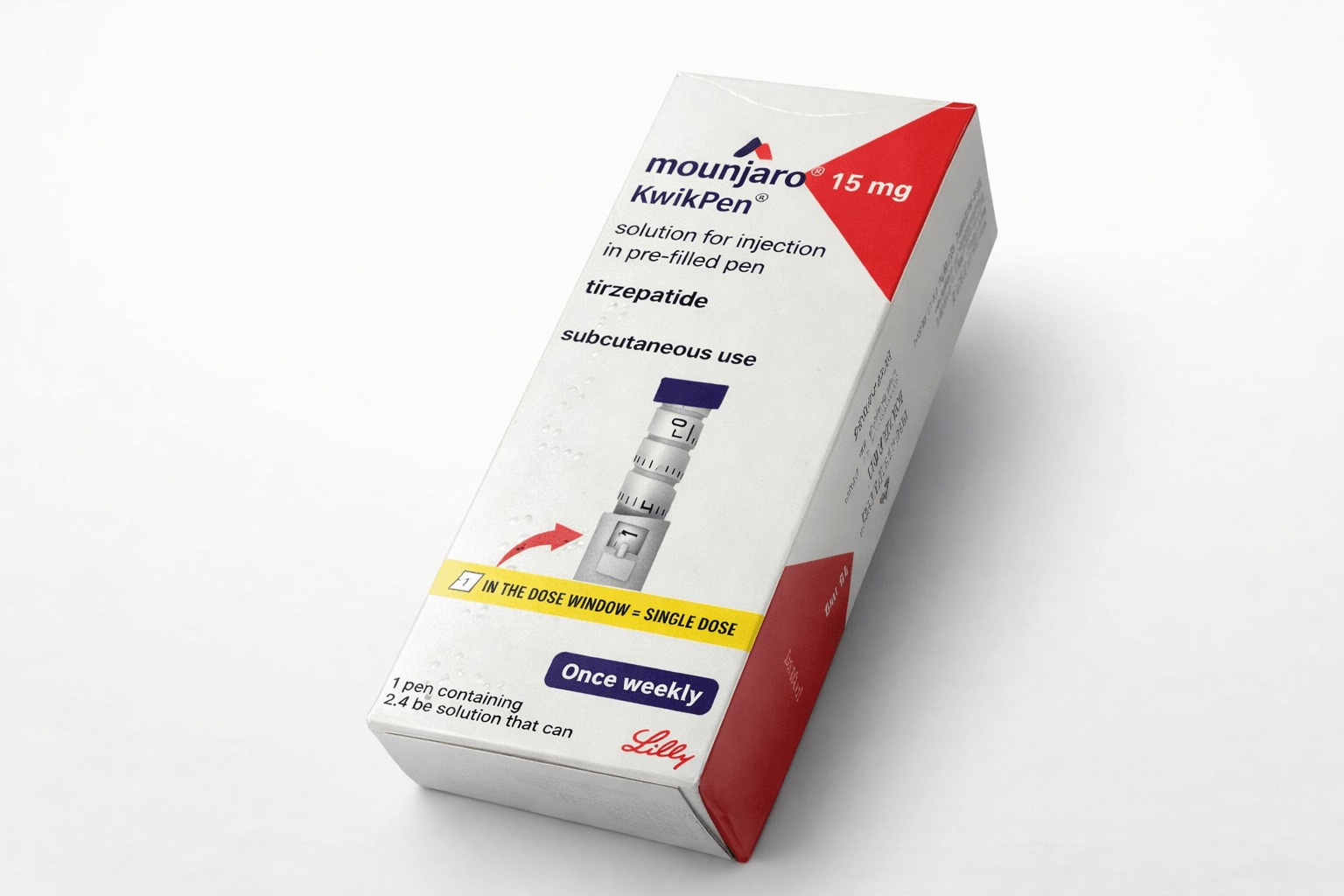
Mounjaro (Tirzepatide) 15mg KwikPen
£266.00
Mounjaro Kwikpen is a once-weekly injection, used to support weight loss and weight management in eligible adult patients.
It works by targeting the GIP and GLP-1 hormones involved in appetite regulation, giving a sense of satiety and is used alongside a healthy diet and lifestyle, following a clinical assessment.
Please Note: You are also required to complete a form at checkout. In addition, a video consultation with our pharmacist may be required.
Categories: Injectable Weight Loss, Online Clinic, Weight Management
Tags: Private Rx
You may be interested in these
What is Mounjaro Kwikpen used for?
Mounjaro Kwikpen is used for weight loss and weight management in adults. It is intended to be used alongside a reduced calorie diet and increased physical activity.
Mounjaro helps reduce appetite and increase feelings of fullness, which may lead to reduced food intake. This can support weight loss and help with maintaining weight over time.
How Mounjaro Kwikpen works
Mounjaro Kwikpen contains the active substance tirzepatide. Tirzepatide is a dual glucose dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. These hormones are involved in the regulation of appetite, food intake and blood sugar levels.
By activating both GIP and GLP-1 receptors, Mounjaro helps to:
- Reduce hunger and appetite
- Increase feelings of satiety after meals
- Slow gastric emptying, helping you feel full for longer
Through these mechanisms, Mounjaro can help reduce overall calorie intake and support weight management when used as prescribed and alongside lifestyle changes.
| Weight | 0.07 kg |
|---|
Always use this medicine exactly as your doctor or pharmacist has told you. Check with your doctor or pharmacist if you are not sure how to use this medicine.
Read the “Instructions for Use” for the pen carefully before using Mounjaro KwikPen.
Each KwikPen contains 4 doses of Mounjaro 2.5mg, 5mg, 7.5mg, 10mg, 12.5mg or 15mg.
The starting dose of Mounjaro is 2.5mg once weekly. After 4 weeks, the dose should be increased to 5mg once weekly. If needed, dose increases can be made in 2.5 mg increments after a minimum of 4 weeks on the current dose. The recommended maintenance doses are 5mg, 10mg and 15mg. The maximum dose is 15mg once weekly.
You can use your pen at any time of the day, with or without meals. You should use it on the same day each week if you can. To help you remember, when to use Mounjaro, you may wish to tick the day of the week when you inject your first dose on the “Instructions for Use” or mark it on a calendar.
You can change the day of your Mounjaro injection, as long as it has been at least 3 days since your last injection. After selecting a new dosing day, continue with once-a-week dosing on that new day.
A small amount of medicine may remain in the pen after all doses have been administered correctly. This is normal and is required for priming the pen. Do not attempt to use any remaining medicine. After the final dose has been given, the pen must be disposed of safely.
Site of Injection
Mounjaro is injected under the skin (subcutaneous injection):
- Stomach area (abdomen) – 5cm away from the belly button in any direction
- Upper leg (thigh)
- Upper arm
It is important to rotate the sit of injection each week to reduce likelihood of skin irritation.
Missed Dose
- If you forget to inject a dose, you have 3 days from your scheduled injection day to take your dose.
- If it has been more than 4 days since your scheduled injection day, skip the missed dose. Then inject your next dose as usual on your scheduled day.
Do not use a double dose to make up for a forgotten dose. The minimum time between two doses must be at least 3 days.
Too much of this medicine may cause low blood sugar (hypoglycaemia). If you use more Mounjaro than you should talk to your doctor immediately.
Instructions for use:
https://www.medicines.org.uk/emc/files/usermanual.15481.pdf
Video for use:
https://www.lilly.com/en-CA/resources/faq/mounjaro-kwikpen/use-kwikpen
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Side Effects
Very Common
- Low blood sugar (Hypoglycaemia) when used with sulphonylurea medications like Glibenclamide, Gliclazide and Glimeprimide or Insulin.
- Nausea
- Diarrhoea
- Vomiting
- Abdominal pain
- Constipation
They are most common when first starting tirzepatide but decrease over time in most patients.
Common
- Decreased appetite
- Low blood sugar (Hypoglycaemia) when used with Metformin, Dapaglifozin, Empaglifozin and Canaglifozin
- Allergic reaction (e.g., rash, itching and eczema)
- Dizziness
- Decreased
- Low blood pressure
- Indigestion
- Bloating
- Burping
- Gas (flatulence)
- Fatigue
- Reflux or heart burn
- Fast pulse
- Injection site reaction (e.g., itching and redness)
- Increased levels of pancreatic enzymes (such as lipase and amylase) in blood
- Increased calcitonin levels in blood
- Gallstones
Symptoms of hypoglycaemia include: Dizziness, sweating, feeling tired, tingling lips, feeling hungry, feeling shaky or trembling, fast or pounding heartbeat and turning pale.
Serious Side Effects
Uncommon
Inflamed pancreas (acute pancreatitis) which could cause severe pain in the stomach and back which does not go away. This is a serious, potentially life-threatening condition.
Symptoms of acute pancreatitis are:
- Severe and persistent pain in the abdomen which might reach through to the back
- Clay like stools
- Nausea and vomiting
If these symptoms are experienced, discontinue treatment and seek medical advice by calling 111 or attending your local A & E department. If acute pancreatitis is confirmed, Mounjaro treatment should not be restarted.
Rare
Severe allergic reactions (e.g. anaphylactic reaction, angioedema). You should get immediate medical help and inform your doctor if you experience symptoms such as:
- Breathing problems
- Rapid swelling of the lips, tongue and/or throat with difficulty swallowing
- A fast heartbeat.
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme, website: www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store. By reporting side effects, you can help provide more information on the safety of this medicine.
Warning & Precautions
Talk to your doctor, pharmacist or nurse before using Mounjaro if:
- you have severe problems with food digestion or food remaining in your stomach for longer than normal (including severe gastroparesis).
- you have ever had pancreatitis (inflammation of the pancreas which may cause severe pain in the stomach and back which does not go away)
- you have a problem with your eyes (diabetic retinopathy or macular oedema).
- you are using a sulphonylurea for your diabetes, as low blood sugar (hypoglycaemia) can occur. Your doctor may need to change your dose of these other medicines to reduce this risk.
Dehydration
When starting treatment with Mounjaro, in some cases you may experience loss of fluids/dehydration, in relation to gastrointestinal side effects and please take precautions to avoid fluid depletion.
Symptoms of dehydration include: dizziness, headache, fatigue, infrequent urination of small amounts, and dry mouth, lips and eyes.
Surgery
If you know that you are due to have surgery where you will be under anaesthesia (sleeping), please consult with your doctor/anaesthetist that you are taking Mounjaro.
Blood glucose levels
If you are using Mounjaro with a sulphonylurea, it is important that you test your blood glucose levels as instructed by your doctor, pharmacist or nurse.
Driving and using machines
It is unlikely that this medicine will affect your ability to drive and use machines. However, if you use Mounjaro in combination with a sulphonylurea or insulin, low blood sugar (hypoglycaemia) may occur which may reduce your ability to concentrate.
Avoid driving or using machines if you get any signs of low blood sugar
Age Restrictions
Mounjaro is supplied to eligible patients aged 18 to 75 years, where treatment is considered clinically appropriate.
Other Medicines and Conditions
Mounjaro delays gastric emptying and has the potential to impact the rate of absorption of concomitantly administered oral medicinal products.
Diabetes and Mounjaro
Please consult your GP if you are taking sulfonylurea medicines for diabetes e.g. glimepiride or glibenclamide or if you are taking insulin – you may experience hypoglycaemia when you use these medicines with Mounjaro.
Therefore, your GP may adjust the dose of the diabetes medicine to prevent you from experiencing hypoglycaemia.
Mounjaro and Poly Cystic Ovary Syndrome
Mounjaro has been shown to regulate menstrual cycles in some individuals, which may increase fertility. If you are not planning a pregnancy at this time, please be aware that this medication could enhance your chances of conception.
Additionally, Mounjaro is a relatively new medication, and there is currently not enough data on its effects during pregnancy or on a developing fetus. For this reason, we strongly advise using an additional barrier method of contraception (such as condoms) alongside your regular form of birth control to reduce the risk of unintended pregnancy.
If your period is more than five days late, we recommend that you see your GP and take a pregnancy test to rule out pregnancy as soon as possible.
If you have any questions or concerns regarding your reproductive health while taking Mounjaro, please do not hesitate to reach out.
Mounjaro and Warfarin, Digoxin and Lithium
The absorption of some medications like Warfarin, Digoxin and Lithium may be affected when taken together with Mounjaro. The manufacturer advises that patients should be closely monitored, as changes in absorption can alter how well the medication works and may affect your blood clotting levels.
For this reason, we recommend that you inform your GP or blood clinic so that they can arrange more frequent blood tests to monitor your INR and ensure your treatment remains safe and effective.
Please keep an up-to-date list of all the medications you are taking and share it with your GP or clinic at your next appointment.
For Warfarin: If you notice any unusual symptoms, such as unexpected bruising, bleeding, or changes in your health, please contact your healthcare provider promptly.
Syncope
Fainting can occur as a psychogenic response to needle injection. This can be accompanied by several neurological signs such as light-headedness, incontinence and tonic-clonic limb movements. Caution should be taken when considering treatment for patients with a history of fainting.
Depression
Depression is a known side effect of Mounjaro. If you start experiencing mood changes, including depression, anxiety or suicidal thoughts, please talk to your GP, as they can help in managing these symptoms.
Thyroid Cancer
Mounjaro may cause tumours in the thyroid, including thyroid cancer. Watch for possible symptoms, such as a lump or swelling in the neck, hoarseness, trouble swallowing, or shortness of breath. If you have any of these symptoms, please speak to your GP straight away.
Please note: If you have a diagnosis of a benign thyroid condition you are at higher risk of developing thyroid cancer.
- Do not use Mounjaro if you or any of your family have ever had a type of thyroid cancer called medullary thyroid carcinoma (MTC).
- Do not use Mounjaro if you have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Do not use Mounjaro if you are allergic to it or any of the ingredients in Mounjaro.
Pregnancy and Breastfeeding
This medicine is not suitable for use in pregnancy and breastfeeding.
Mounjaro Kwikpen should not be used during pregnancy as the effects of this medicine on an unborn child are not known.
Mounjaro should not be used during pregnancy or in women of child bearing potential not using contraception. If you wish to become pregnant, you must stop using Mounjaro at least 1 month before a planned pregnancy due to the long half-life of Mounjaro.
If you become pregnant when using Mounjaro, you should contact your GP and treatment provider.
If you take oral contraceptives, should consider also using a barrier method of contraception (e.g., a condom) or switching to a non-oral contraceptive method for 4 weeks after starting Mounjaro and for 4 weeks after each increase in dose.
Patient information Leaflet
https://www.medicines.org.uk/emc/files/pil.15481.pdf
Ingredients
- Active Ingredients: Tirzepatide
- Other Ingredients: Sodium phosphate dibasic heptahydrate, Sodium chloride, Concentrated hydrochloric acid, and sodium hydroxide (for pH adjustment), Glycerol, Phenol, Benzyl Alcohol and Water for injections
Product Information
- Brand Name: Mounjaro Kwikpen
- Route of Administration: Subcutaneous
- Legal Classification: Prescription Only Medication (POM)
- Strength: 2.5mg, 5mg, 7.5mg, 10mg, 12.5mg and 15mg
- Dosage Form: Solution for injection
Storage
- Keep this medicine out of the sight and reach of children.
- Store in original package in order to protect from light.
- Mounjaro should be stored in a refrigerator (2°C – 8°C).
- Do not freeze. Keep away from the freezer compartment.
- After first use, Mounjaro may be stored unrefrigerated for up to 30 days at a temperature not above 30°C and then the pre-filled pen must be discarded.
- Do not use this medicine if you notice that the pen is damaged, or the medicine is cloudy, discoloured or has particles in it.
Disposal
- Do not throw away any medicines via wastewater or household waste.
- Please discard the pre-filled pen and sharps needles into a sharps box.
Advice & Helpful Links
It is vital that Mounjaro is used as part of an integrated approach to weight management, with the appropriate support, counselling and behavioural strategies available to you. Mounjaro should be used alongside a balanced diet and healthy lifestyle to support weight loss and overall wellbeing.
Protein intake: Including enough protein (100g of protein) in your diet may help maintain muscle mass and increase feelings of fullness. Protein-rich foods include lean meats, fish, eggs, dairy, legumes, and plant-based alternatives.
Total Daily Energy Expenditure (TDEE): Weight loss occurs when calorie intake is lower than energy expenditure. Understanding your estimated daily calorie needs (TDEE) may help with planning meals and managing portion sizes. Reducing calorie intake should be done in a safe and sustainable way.
Please adhere to the dietary recommendations that are given. This includes a nutritionally balanced, mildly hypocaloric diet of 500-600 calorie deficit, in addition to a diet that is rich in fruits, vegetables and protein.
Water intake: Staying well hydrated is important, 2-3L a day, particularly while using appetite-reducing medicines. Drinking water regularly throughout the day may help support digestion and reduce the risk of dehydration, especially if you experience reduced appetite or gastrointestinal side effects.
Exercise: Gradually add more activity to your routine. Even small increases in resistance training or cardio can make a difference.
Regular physical activity, adequate sleep, and consistent eating patterns can support weight management. Making gradual, sustainable changes can help aid with long term maintenance.